By the EHR Association COVID-19 Task Force
In December, two COVID-19 vaccines received authorization from the FDA, and the federal government began distribution to the states almost immediately. Millions of Americans have already received their first dose – many their second – and millions more will be vaccinated in the coming weeks and months. While social distancing, frequent hand washing, and face masks remain vital tools in limiting spread of coronavirus, we can increasingly see our way to a full return to hugs and handshakes, in-person meetings, travel, pubs and parties, concerts and classrooms.
As we anxiously await a return to our old way of life, public health experts, policymakers, and the public are watching the CDC vaccine data tracker, updated every evening with the latest numbers, including:
- How many vaccine doses have been distributed to-date?
- How many vaccines have been administered?
- How many people have received their first dose?
- How many people have gotten a second dose?
- Which vaccine is being administered?
But how does the CDC get all that data? The answer varies, but it’s made possible by technology, and electronic health records (EHRs) have been a key player from the beginning.
Here are five ways that EHRs and the EHR Association are playing important roles in vaccine administration and data collection in the United States.
1. Communication and Coordination
As Operation Warp Speed evolved, EHRA and EHR developers communicated regularly with HHS, CDC, the American Immunization Registry Association (AIRA), and other stakeholders.
The EHR Association played a leadership role in facilitating these conversations, allowing our member companies to directly share their questions, concerns and feedback with the CDC and other stakeholders managing nationwide data collection efforts. Our members have been able to provide the developer perspective, as well as relay the challenges our provider customers have expressed to us. We’ve been able to help CDC and other public health agencies effectively collect the data they need using EHR capabilities, while minimizing burden to already overwhelmed healthcare providers.
2. Identifying and Incorporating Data Elements
Before the first vaccines were even approved, government health agencies and stakeholder associations, including EHRA, worked together to identify and understand the data tracking needs that would be required to report and track vaccine administration. EHR developers were then able to incorporate new elements into their software – such as vaccine codes – ahead of the vaccines being made available. Vaccine codes are important, not only to track vaccine supply, but to ensure that people get the same vaccine for the second dose, at the right interval. Many EHRs are able to manage scheduling the second dose and sending reminders to patients.
Still, data collection has not been without challenges. EHRs have long been used to support vaccine administration, but as the COVID vaccinations started, new data requirements emerged that had not been included in immunization reporting before. The widespread need for vaccinations revealed emerging problems of equity and access, and collecting certain data took on renewed importance: race and ethnicity, priority groups, and other social determinants of health.
Additionally, the accelerated vaccine development and deployment process resulted in gaps in available data: e.g., the barcode on vaccine vials initially did not include expiration dates. Variations in state reporting requirements created further challenges. EHRA members collaborated closely with HHS, CDC, AIRA, and states to identify needs and opportunities to align data requirements across jurisdictions, while sharing best practices and ways to accelerate support
3. Streamlined Public Health Data Collection & Reporting
As the U.S. raced toward a vaccine, EHR developers collaborated with CDC and state public health registries to ensure that when the time came, EHR technology would be able to effectively and efficiently exchange data on who was receiving the vaccine. The federal government has set an expectation that providers submit vaccine administration data daily, and the goal is for COVID-19 vaccines to be documented and administered in, and reported on, by the EHR the same way as a flu vaccine, within the workflow and without added burden.
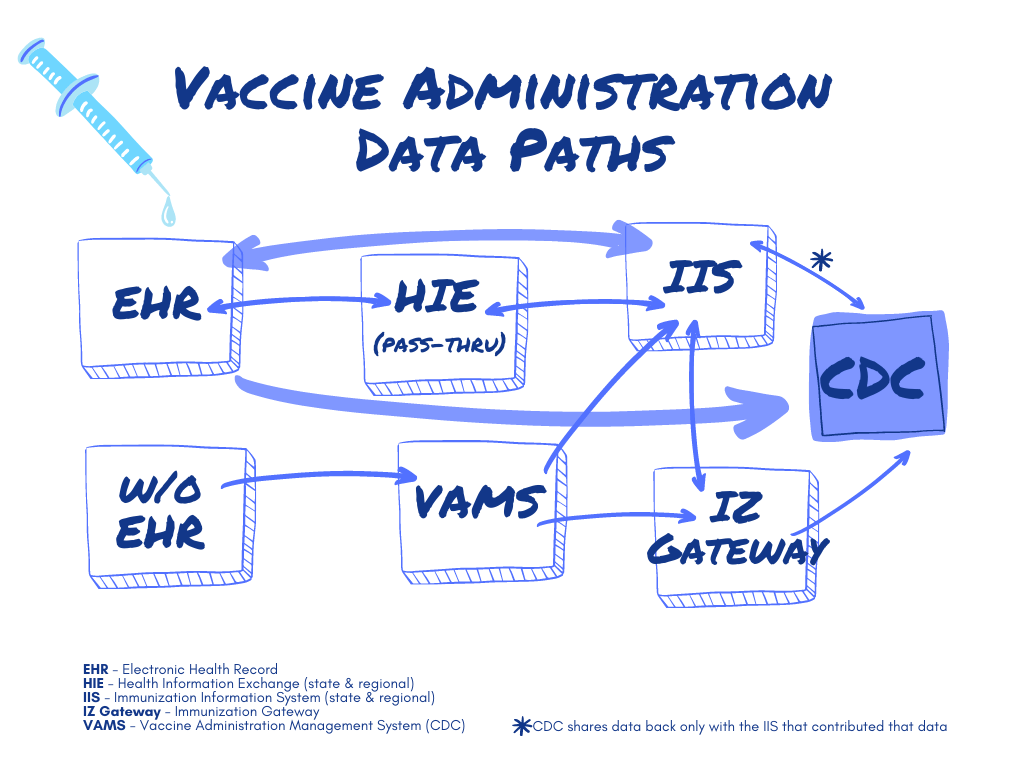
But as the graphic below shows, reporting vaccine administration in the U.S. is not as simple as one might expect. Most vaccine data flows through one of the country’s 64 Immunization Information Services (IIS) – sometimes first collected by a health information exchange (HIE) – and from there continues on a path to the CDC.
Public health researchers are able to comb de-identified patient records received through the EHRs to identify areas of community spread of COVID-19, understand vaccine effectiveness and reinfection potential, spot mutated virus strains, and manage prioritization, eligibility, and distribution through the vaccine administration process.
The CDC/FDA Vaccine Adverse Event Reporting System (VAERS) and V-Safe program enables individuals to report on any reactions, enabling CDC and FDA tor to detect unusual or unexpected patterns that may indicate a safety concern with a vaccine, and is especially valuable for recently-authorized vaccines, such as for COVID-19.
4. Safer Immunizations Through Interoperability
EHRs support the ability to query immunization registries, centralized hubs of vaccination records. This enables providers to have a more complete view of a patient’s immunization record. Immunization registries connected via a centralized architecture called the IZ Gateway are able to share patient records across jurisdictions, thus expanding a provider’s access to important vaccination information. This is particularly important with two-dose regimens such as the Pfizer-BioNTech and Moderna COVID-19 vaccines, in cases where the first vaccination was administered in one jurisdiction, but the second needs to be administered by a provider in another jurisdiction. EHRs can use this information to schedule the second vaccination at the right time, regardless of where the patient went before. An EHR connection to an IIS registry or the IZ Gateway can also provide information about vaccination status when a patient presents for an elective procedure, helping make sure that clinicians and staff are prepared with proper precautions and that resources such as PPE are used effectively.
Accurate identification of patients is one of the most difficult operational issues during a public health emergency, including gathering patient demographic information, and ensuring information remains attached to the correct patients so that second doses can be appropriately timed. Patient identification is a key component of nationwide interoperability, and the pandemic has highlighted this urgent need to match the right patient with the right health data. EHR developers have long advocated for a national strategy for patient identification in order to enable improvements in healthcare quality, affordability and outcomes.
5. Educating and Supporting Providers
EHR developers, through discussions with CDC and state and regional public health agencies, are intensely focused on staying abreast of government reporting requirements, to help their customers remain in compliance with the latest expectations.
As vaccine approvals moved closer, EHR developers added new tools and rolled out mobile applications to support mass vaccination sites for both a provider’s current and new patients. Developers have worked with healthcare organizations and providers every day to coach them on how to use these new COVID-19 related tools in their EHR to accurately document the information needed for public health data collection efforts. These elements can be incorporated into provider workflows, automating collection and transmission of required data to regional immunization registries that ultimately share information with the CDC. From documentation to webinars to one-on-one training conversations, developers are going to extraordinary lengths in an extraordinary time to support the people that rely on their software.
As we noted in a recent blog post about the role of technology in public health data collection, COVID-19 reporting has the opportunity to be streamlined. Rather than relying on spreadsheets and faxes, thanks to EHRs more than 90% of hospitals are sending digital immunization reports, 88% are submitting electronic lab results (ELR), and 76% are electronically providing syndromic surveillance data to public health agencies.
EHRA and EHR developers are proud of our work to support the heroic efforts of healthcare providers and public health agencies during this devastating pandemic.